|
THIN
LAYER CHROMATOGRAPHY (TLC)
How Everything
Started.
I believe that they did not
start with TLC as TENDER, LOVE and CARE but one thing that the Scientists
called Thin Layer Chromatography. Everything began when one had an idea to
coat a glass plate with silica gel and then after apply a small drop of sample
one solvent was used to run up the glass plate by capillarity.
Look carefully each figure below and You will be able to understand such
technique that is simple and do not needs so much skills.
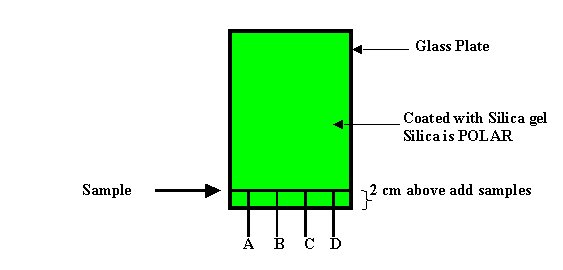
It was created a special
chamber that was called the TLC CHAMBER, which was improved a lot after many
experiences with a lot mistakes and few success. I could talk a lot about such
improvements but this is only an idea how HPLC started.
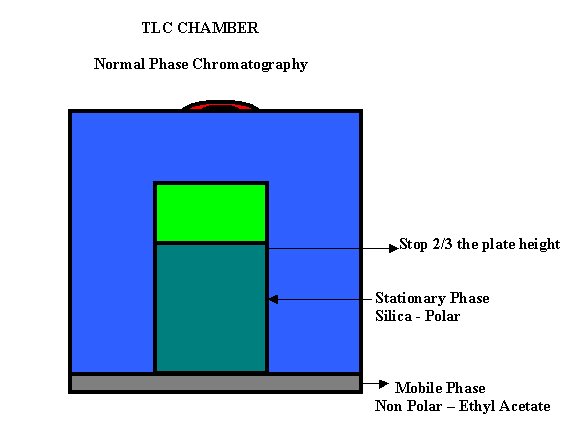
The solvent by capillarity
goes up In Thin Layer Chromatography the solvent or more known as Mobile
Phase is a Non Polar substance such as Ethyl Acetate. The Stationary Phase is
Polar because it consist of Silica that is Polar.
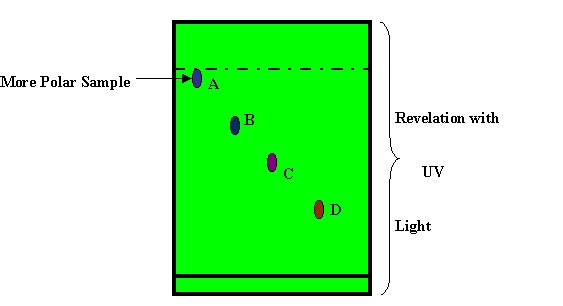
Finally the silica plate with
samples is take off from the TLC Chamber and dry at room temperature.
Note: Such Operation Must Be Done in a Fume Hood to Avoid the Chemical Fumes
that are Health Hazardous.
The revelation is done in generall with Ultra Violet light because most the
Pharma Products contain Amines (NH2).
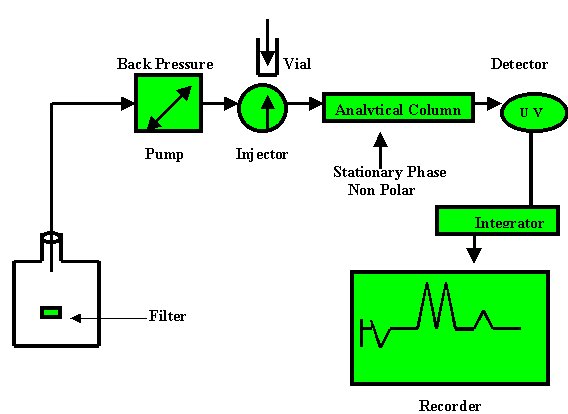
REVERSE PHASE CHROMATOGRAPHY (RPC)
The name says a lot. Reverse
Phase Chromatography because now the Stationary Phase is Non Polar and the
Mobile Phase is Polar or Moderately Polar.
Let's to analyse
the Mobile Phase:
Aqueous
+ Organic
H2O
that is highly polar but weaker solvent.
Organic modifier that can be Methanol (MeOH); Acetonitrile (ACN) and
Tetrahydrofuran (THF).
Once the Polarity of Solvent Decreases, the Solvent Strength Increases.
Consequently 52% Methanol = 48% Acetonitrile = 32% Tetrahydrofuran.

I will keep going on with
my explanations later.
Index
|
 Welcome to
HPLC
site!
Welcome to
HPLC
site!